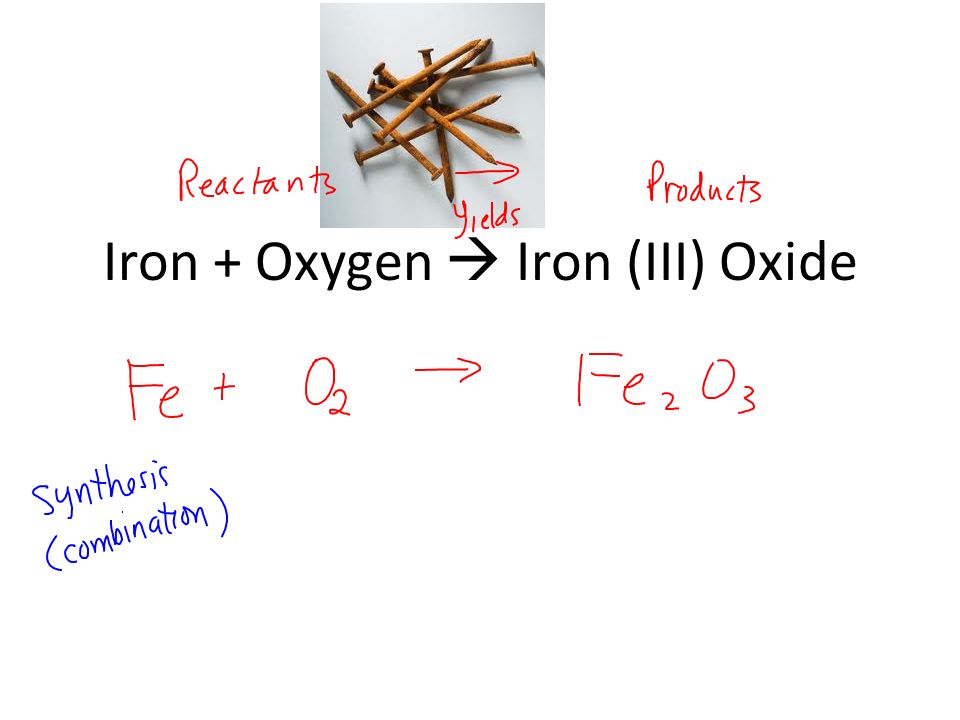
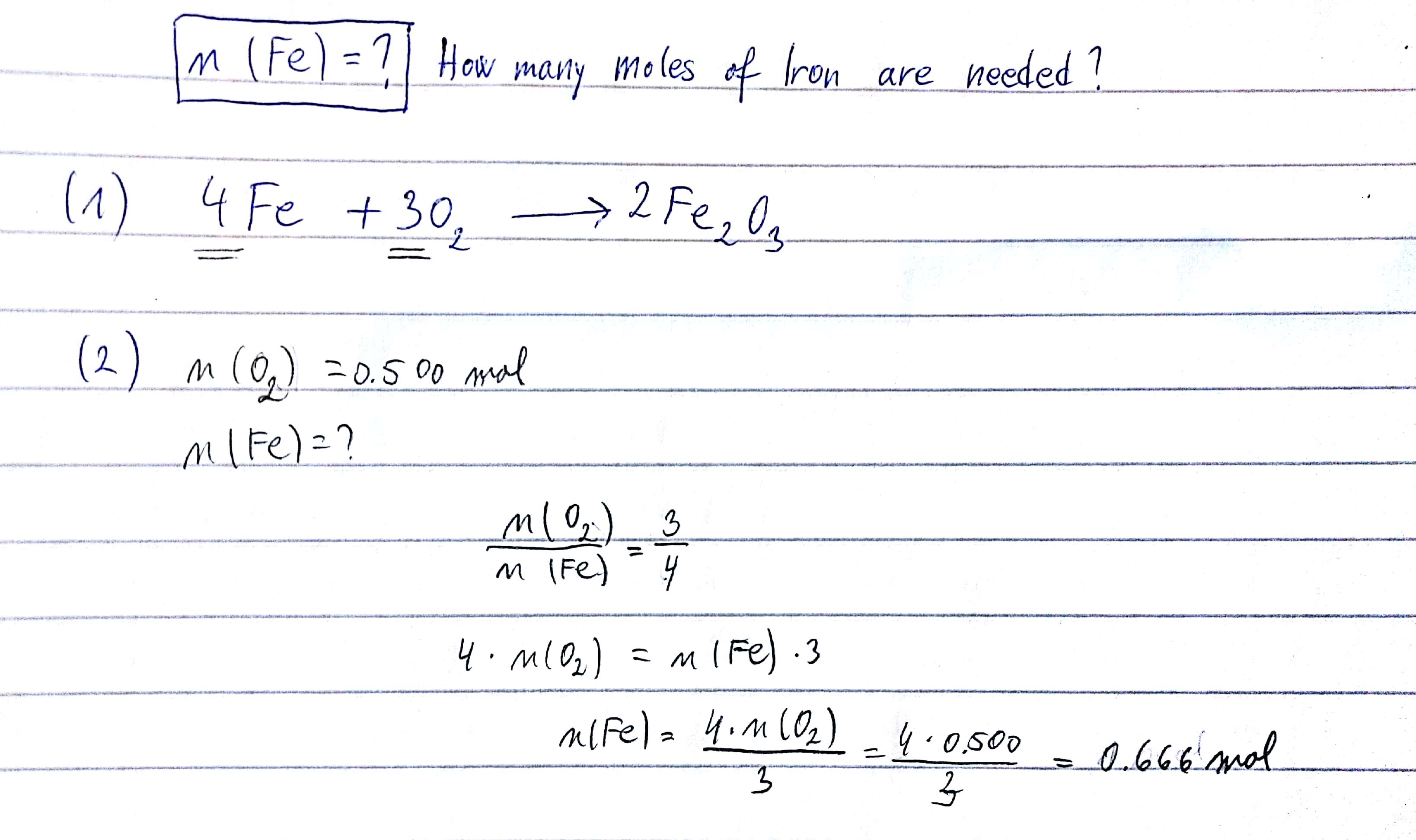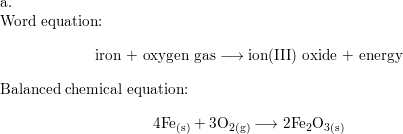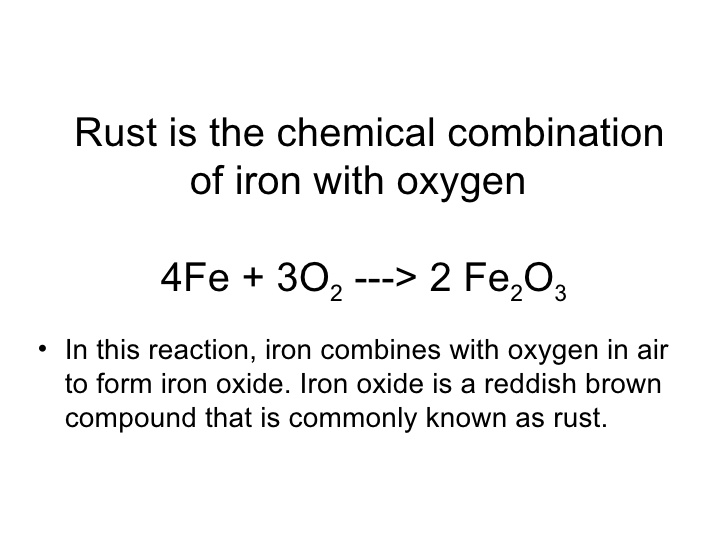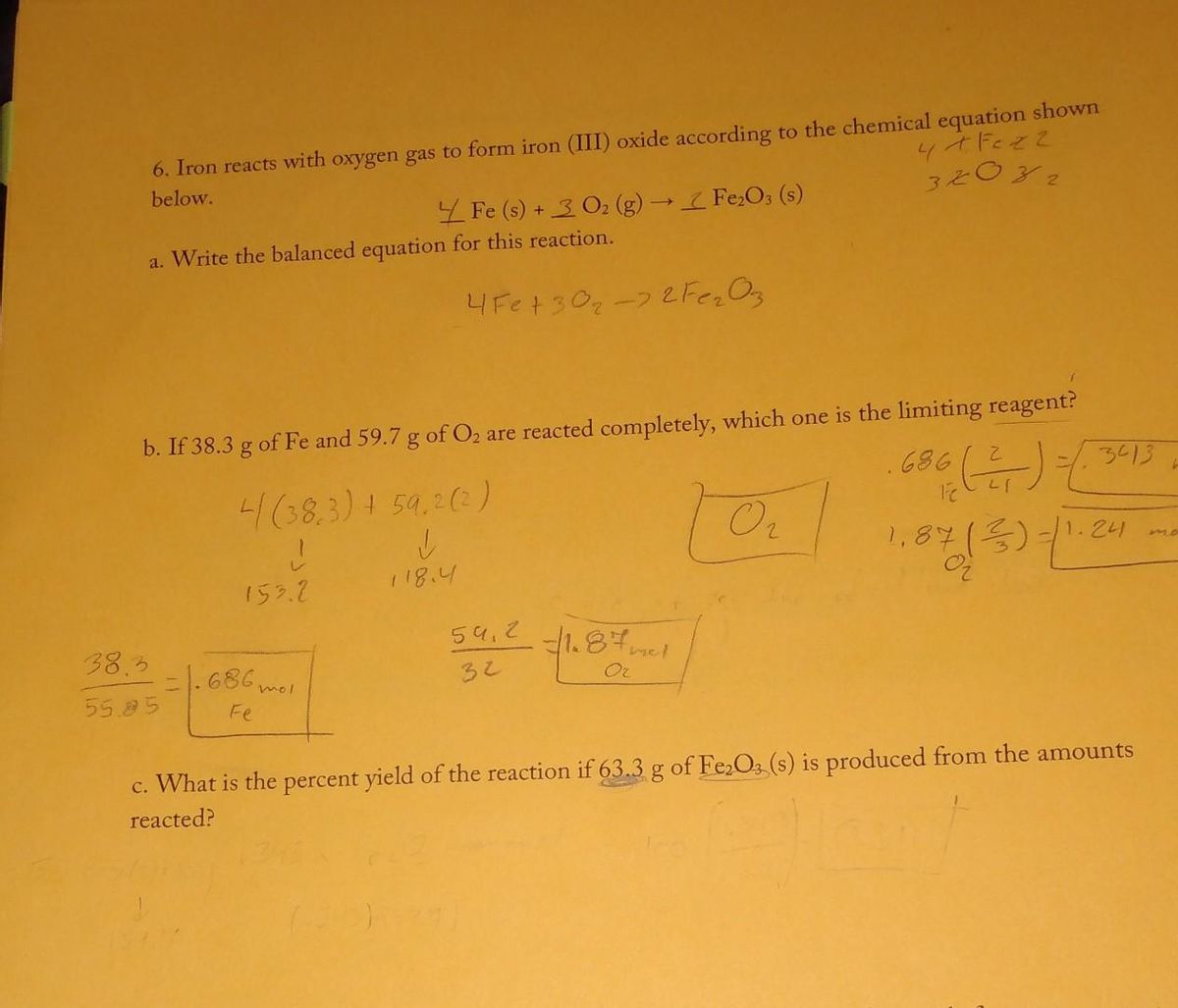⚗️Iron (Fe) reacts with oxygen gas (O2) to form rust (Fe2O3). Balance the equation below by writing - Brainly.com

Simplified chemical reactions between iron and oxygen within the haem... | Download Scientific Diagram

What is oxidation of iron in inorganic chemistry? | by KAKALI GHOSH , Teacher,blogger. M.Sc chemistry. | Medium
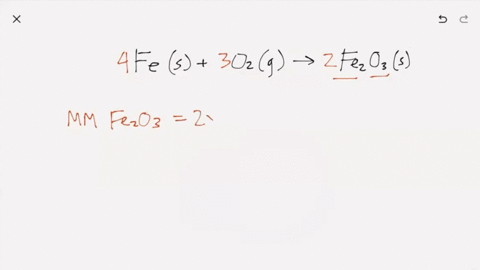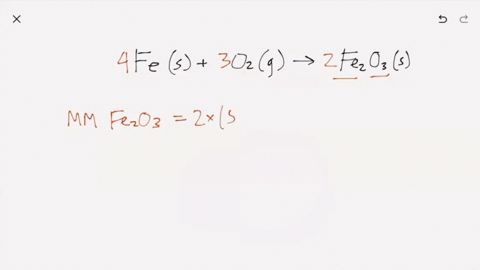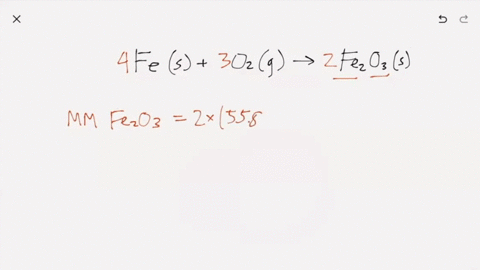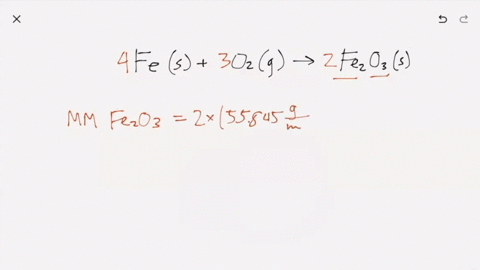
SOLVED:Iron metal reacts with oxygen to give iron(III) oxide, Fe2 O3 (a) Write a balanced equation for the reaction. (b) If an ordinary iron nail (assumed to be pure iron) has a